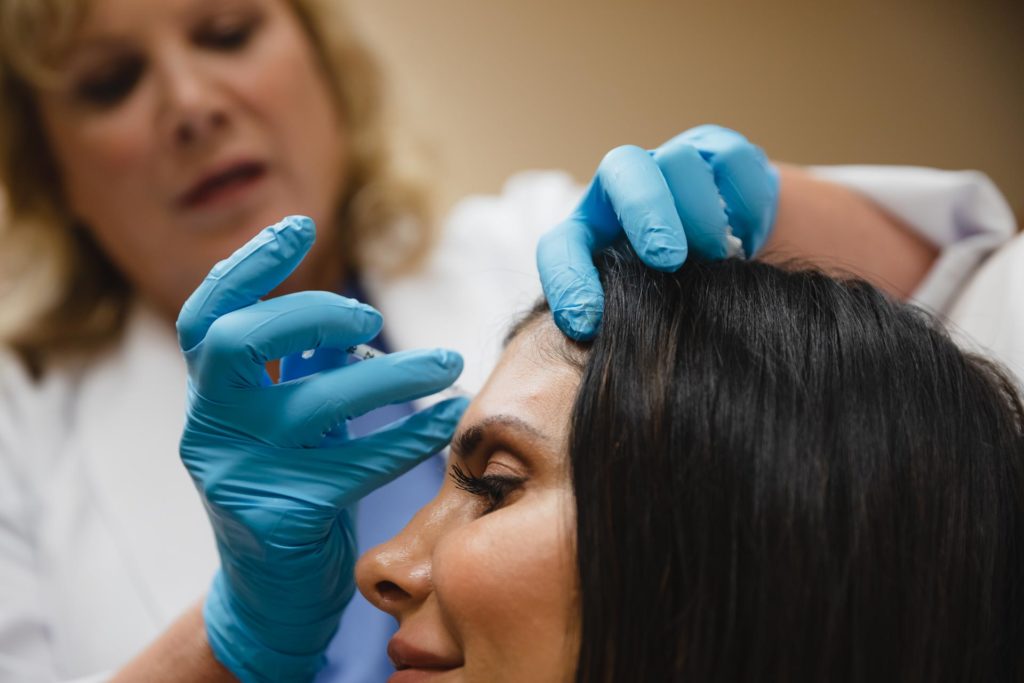
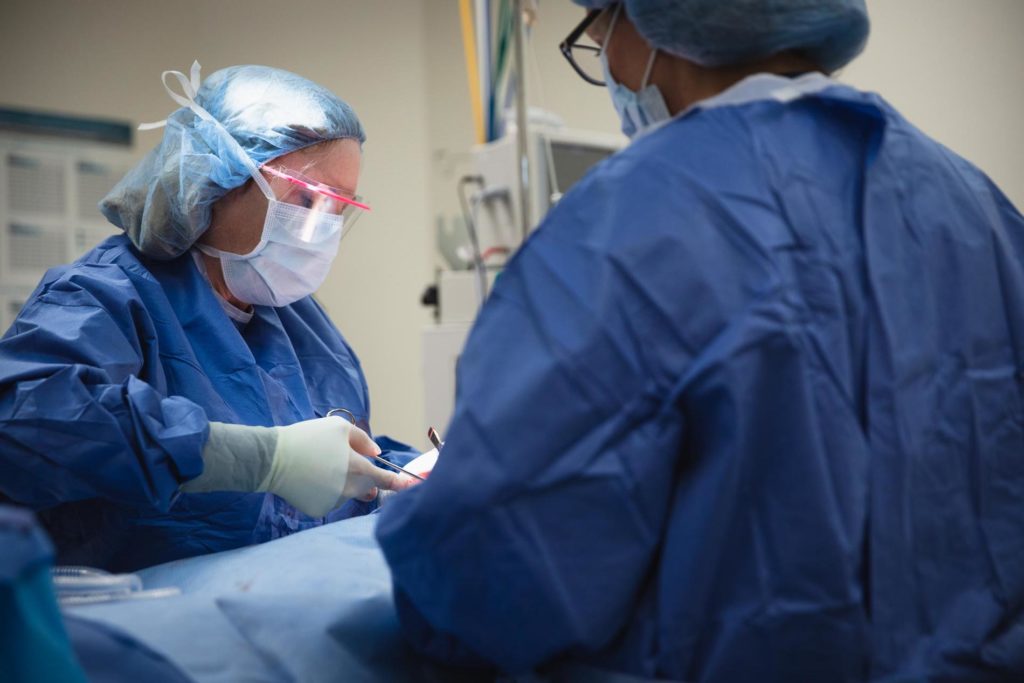
Turkle & Associates is the premier provider of plastic surgery in Indiana.
Passion and experience drive our actions every day, which means you’ll get the best experience every time. Learn more about our process for plastic surgery in Indianapolis at Turkle & Associates Cosmetic & Reconstructive Surgery.

Your partner in aesthetic wellness
Wanting to look your best is something to be celebrated. When you make the decision to pursue plastic surgery in Indianapolis, you want an expert team to help you know what to expect. Dr. Janet Turkle and her team are known for their exceptional patient care and knowledgeable attention.
From your initial phone call to the final follow-up, we’re here to make your plastic surgery experience an excellent one. When you’re looking for the best plastic surgeon in Indianapolis, you want an expert surgeon committed to their patient’s well-being and aesthetic goals. And that’s exactly what you’ll find with Dr. Turkle.
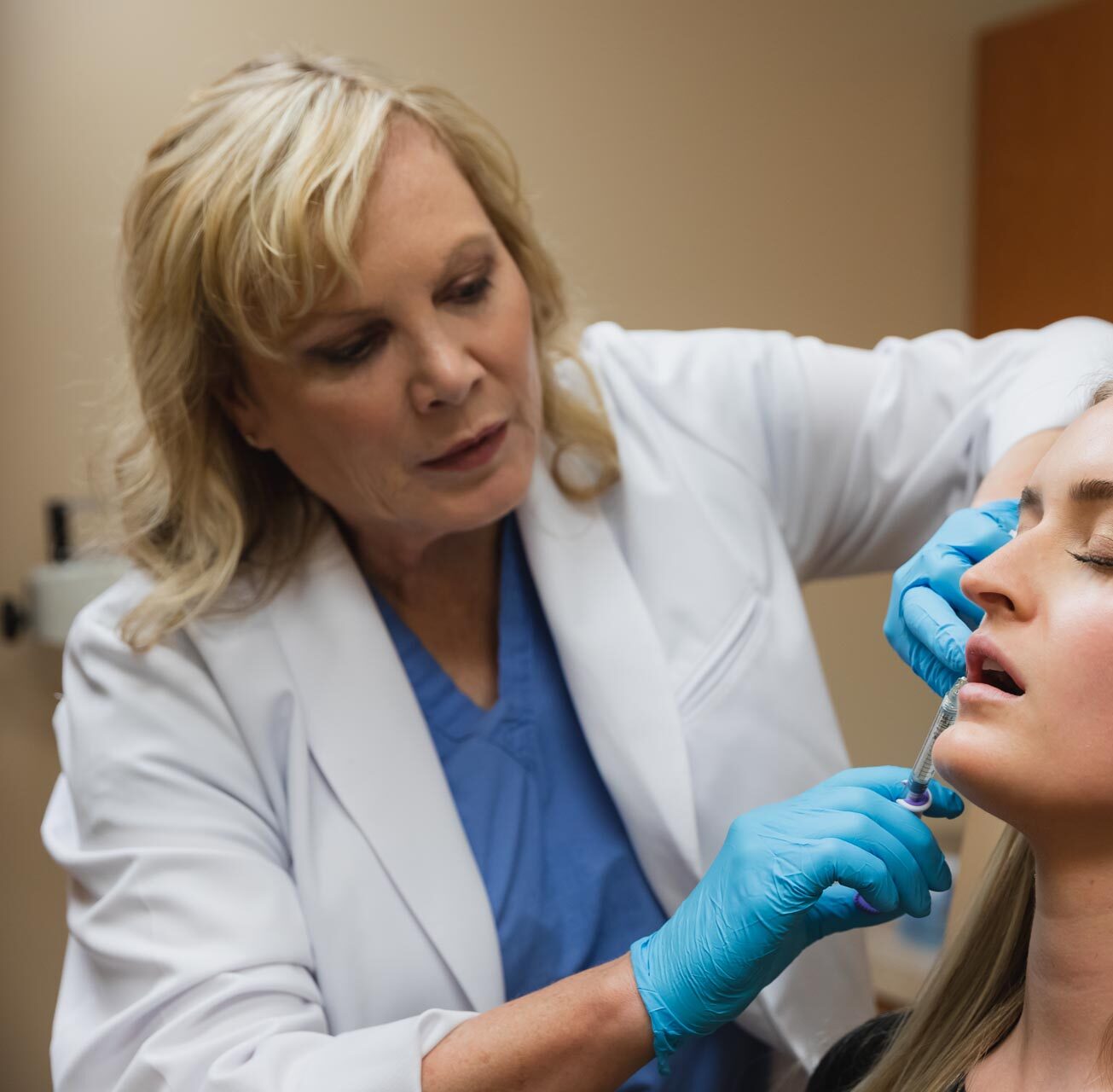
About Dr. Turkle
Dr. Janet Turkle is dual-board certified by the American Board of Surgery and the American Board of Plastic Surgery. She is proud to have been named a Top Doc for Cosmetic Surgery in Indianapolis as well as for Plastic and Reconstructive Surgery.
Together with her team, Dr. Turkle is committed to helping our patients actualize the best versions of themselves, restoring their self-confidence and comfort in their appearance.

It all starts with a conversation
Our team creates personalized treatment plans for all of our patients. We take the consultation process very seriously as our chance to get to know you, your goals, and how we can work together to achieve them. We encourage you to ask questions and ensure you are comfortable with our staff and facilities.
Turkle & Associates guides our patients through options for plastic surgery in Indianapolis, as well as incredible, non-invasive medical spa treatments. You tell us your goals and we use our expertise to meet them.
Look good. Feel your best.
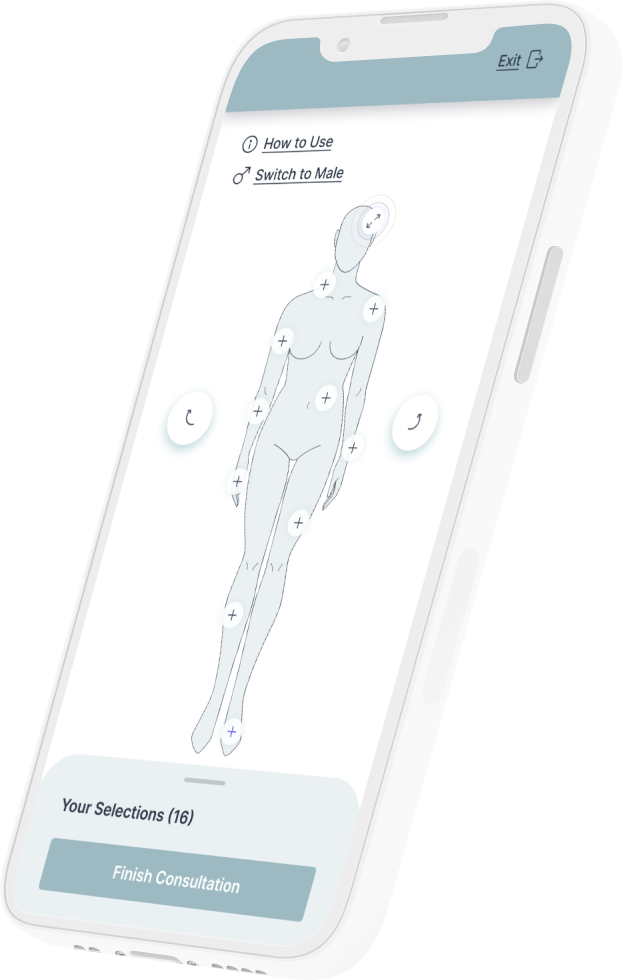
Tell us your concerns using our instant online virtual consultation to discover which procedures may be best for you.

Our partner med spa, Phases Skin Care and Laser Center
We’re committed to the comprehensive well-being of our patients and offer a suite of non-surgical cosmetic treatments through our medical spa Phases Skin Care and Laser Center. Whether you’re interested in less invasive methods or simply want to protect your surgical results in the long term, we have treatments available.
No matter what your aesthetic goals are, we can help you reach them, through medical-grade skincare products, an extensive array of laser technologies, and innovative body contouring treatments.
Popular Products
Our main office is located at 11455 North Meridian Street, Suite 150, Carmel, Indiana 46032.
Monday 8AM–5PM
Tuesday 8AM–5PM
Wednesday 8AM–5PM
Thursday 8AM–5PM
Friday 8AM–4PM
Saturday Closed
Sunday Closed
You can call our office at 317.848.0001 or you can request an appointment online.
For patients interested in plastic surgery in Indianapolis, we always start with a consultation to ensure candidacy and build your treatment plan.
News


Don't forget your sunscreen this summer!☀️
Take advantage of our 20% off sale for ALL SUNSCREENS this month of May!
#sunscreen #wearsunscreen #mineralsunscreen #sunscreencream #sunscreens #beautysunscreen #sunscreenspf #alastinskincare #alastin #carmelindiana #carmelin #skincancerawarenessmonth #skincancerawareness #skincancerprevention #spf #sunscreeneveryday #sunscreenalways #sunscreenprotection #summertime
Whether you want a little bit more fullness or to correct the volume loss that happens over time, we have the solution for you!
Ready for a little lip plump? Click the link in our bio! 💋
#lipfiller #lipfillerbeforeandafter #lipinjections #lipvolume #lipplump #lips #carmelindiana #carmel #indy #indylips
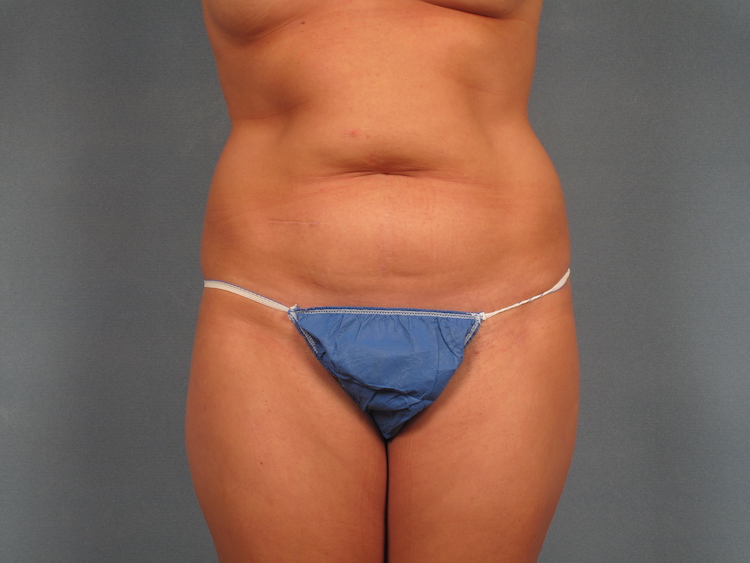
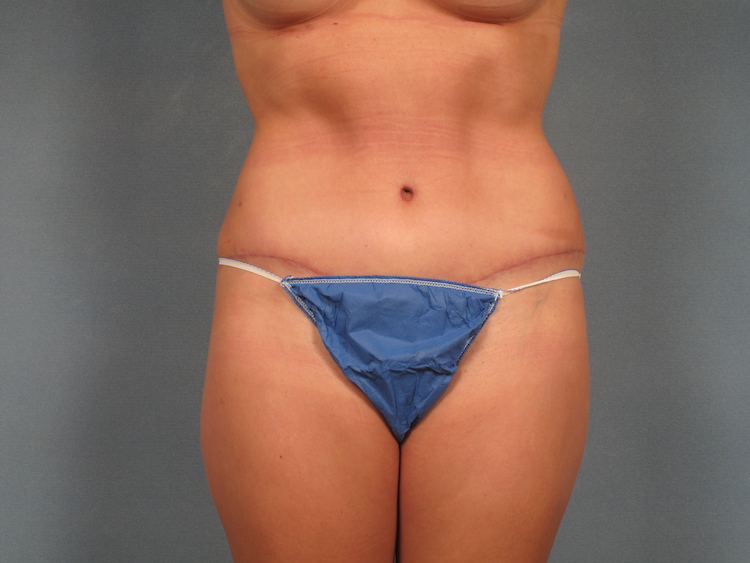
Seeking clarity on Liposuction and Tummy Tuck? Listen closely!
Let Dr. Tukle guide you toward your desired results.
Want to learn more? Clck the link in our bio to book your consultation.
#liposuction #tummytuck #aestheticsurgery #liposuctionsurgery #carmel #carmelindiana #visitcarmel #carmelin
Achieve a flawless complexion with the help of Hydrafacial.
Hydrafacial is a fast, gentle skin treatment that cleanses, exfoliates, hydrates, and protects the skin. It improves the appearance of fine lines, wrinkles, acne, and congested pores, takes 30 minutes, and has no downtime.
Book your Hydrafacial appointment today!
#hydrafacial #hydrafacials #hydrafacialmd #hydrafacialist #hydrafacialglow #hydrafacialtreatment #carmelindianapolis #carmel #carmelindiana #visitcarmel #carmelmoms
Error: API requests are being delayed. New posts will not be retrieved for at least 5 minutes.
There may be an issue with the Instagram access token that you are using. Your server might also be unable to connect to Instagram at this time.
Click here to troubleshoot.